- Key takeaways
- Part 1. What is a sodium-ion battery?
- Part 2. How sodium-ion batteries work
- Part 3. Sodium-ion battery vs lithium-ion battery
- Part 4. Sodium-ion battery construction
- Part 5. How sodium-ion batteries are made
- Part 6. Sodium-ion batteries life
- Part 7. Performance characteristics of sodium-ion batteries
- Part 8. Where sodium-ion batteries make the most sense
- Part 9. Challenges of sodium-ion battery technology
- Part 10. Will sodium-ion battery replace lithium-ion battery?
- Part 11. The future of sodium-ion batteries
- Part 12. FAQs
Lithium-ion batteries dominate today’s energy storage market. But if you’re working in battery sourcing, product development, or energy storage planning, you’ve probably noticed something new gaining attention: sodium-ion batteries.
They’re not just another lab experiment anymore. Manufacturers are already commercializing sodium-ion technology for energy storage systems, low-cost EVs, and backup power. And if you’re evaluating long-term battery strategies, understanding how sodium-ion batteries work — and how they’re made — can help you make smarter decisions.
Let’s walk through the technology in a practical, engineering-focused way.
Key takeaways
- Sodium-ion batteries work similarly to lithium-ion batteries but use sodium ions instead of lithium ions
- They offer lower cost, better safety, and improved low-temperature performance
- Sodium-ion battery construction includes cathode, anode, electrolyte, separator, and current collectors
- Manufacturing processes are largely compatible with lithium-ion production lines
- Sodium-ion batteries are ideal for energy storage, backup power, and cost-sensitive applications
Part 1. What is a sodium-ion battery?
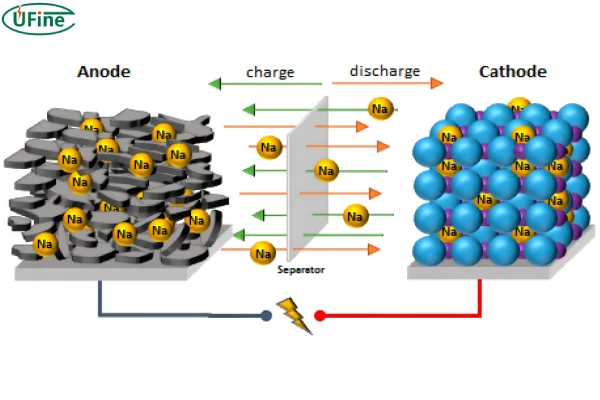
A sodium-ion battery is a rechargeable battery that uses sodium ions (Na⁺) as charge carriers. During charging and discharging, sodium ions move between the cathode and anode — just like lithium ions in lithium-ion batteries.
So why sodium?
Because sodium is:
- Abundant in nature
- Low cost
- Widely available across regions
- Less sensitive to supply chain risks
Unlike lithium, sodium can be extracted from sea salt and abundant minerals, making it attractive for large-scale energy storage.
Part 2. How sodium-ion batteries work
The working principle of a sodium-ion battery is based on reversible sodium-ion intercalation.
Here’s what happens:
- During charging, sodium ions move from cathode to anode
- During discharge, sodium ions return to the cathode
- Electrons flow through the external circuit to provide power
This process is similar to lithium-ion batteries, which is why sodium-ion technology can reuse much of the existing lithium-ion manufacturing infrastructure.
However, sodium ions are larger and heavier than lithium ions, which affects energy density and material selection. Engineers must carefully design electrode materials to maintain performance and cycle life.
To better understand sodium-ion technology, it’s helpful to review how a battery works first.
Part 3. Sodium-ion battery vs lithium-ion battery
To understand sodium-ion battery technology, it helps to compare it directly with lithium-ion.
| Feature | Sodium-ion battery | Lithium-ion battery |
|---|---|---|
| Raw material cost | Lower | Higher |
| Energy density | Lower | Higher |
| Safety | Better thermal stability | Moderate |
| Low-temperature performance | Good | Moderate |
| Resource availability | Very abundant | Limited |
| Manufacturing compatibility | High | — |
| Best applications | Energy storage, backup | EVs, electronics |
From an engineering perspective, sodium-ion batteries are not designed to replace lithium-ion completely. Instead, they complement lithium-ion in areas where cost and safety matter more than energy density.
Part 4. Sodium-ion battery construction
A typical sodium-ion battery construction includes the following main components:
- Cathode
- Anode
- Electrolyte
- Separator
- Current collectors
1 Cathode materials
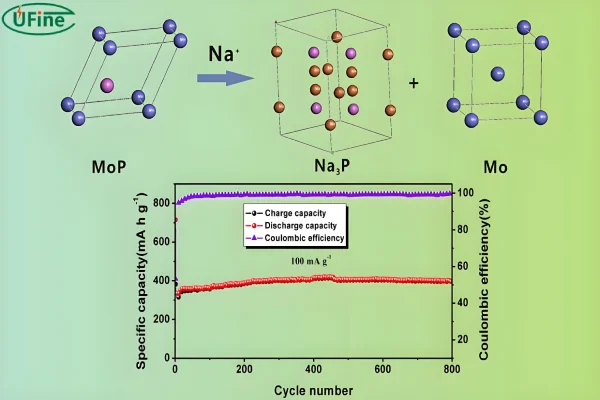
Common sodium-ion cathode materials include:
- Layered transition metal oxides
- Polyanionic compounds (NaFePO₄, Na₃V₂(PO₄)₃)
- Prussian blue analogs
Among these, Prussian blue materials are gaining attention because of low cost and fast sodium diffusion.
2 Anode materials
Unlike lithium-ion batteries that use graphite, sodium-ion batteries often use:
- Hard carbon
- Soft carbon
- Sodium titanate
Hard carbon is currently the most widely used anode material due to good stability and moderate capacity.
3 Electrolyte
Electrolytes in sodium-ion batteries are similar to lithium-ion systems:
- Sodium salt (NaPF₆, NaClO₄)
- Organic solvent (EC, PC, DEC)
These components allow sodium ion transport between electrodes.
Part 5. How sodium-ion batteries are made
One of the biggest advantages of sodium-ion batteries is that manufacturing is very similar to lithium-ion production. This reduces investment costs and speeds up commercialization.
The basic manufacturing process includes:
- Electrode material preparation
- Slurry mixing
- Coating on current collectors
- Drying and calendaring
- Electrode cutting
- Cell assembly
- Electrolyte filling
- Formation and aging
Because the production steps are similar, many lithium-ion factories can adapt existing lines for sodium-ion battery production.
This compatibility is one reason why sodium-ion technology is progressing faster than expected.
If you’re curious about how batteries are manufactured at scale, check our battery production process for a step-by-step overview.
Part 6. Sodium-ion batteries life
Sodium-ion batteries are designed for long-term stability, especially in stationary energy storage applications. While their energy density is lower than lithium-ion batteries, their cycle life and durability are becoming increasingly competitive.
Most sodium-ion batteries typically deliver 2,000 to 5,000 charge cycles, depending on materials, temperature, and depth of discharge. Some newer chemistries, particularly those using Prussian blue cathodes and hard carbon anodes, are already reaching even higher cycle performance in controlled conditions.
In real-world applications, this usually means:
- 5–10 years for moderate-use applications
- 8–15 years for energy storage systems
- Longer lifespan when used at partial depth of discharge
Another advantage is that sodium-ion batteries often show better tolerance to full discharge and storage at low voltage, which helps reduce degradation during long idle periods.
However, battery life still depends heavily on:
- Operating temperature
- Charging speed
- Depth of discharge
- Battery management system (BMS) design
If you’re comparing different battery technologies, you may also want to read lithium battery lifespan for reference.
Part 7. Performance characteristics of sodium-ion batteries
| Parameter | Typical range |
|---|---|
| Energy density | 100–160 Wh/kg |
| Cycle life | 2000–5000 cycles |
| Operating temperature | -20°C to 60°C |
| Charging speed | Moderate to fast |
| Safety | High thermal stability |
While the energy density is lower than lithium-ion, sodium-ion batteries often outperform in cold environments and offer improved safety.
Sodium-ion Battery Pros
- Abundance and Low Cost: Sodium is one of the most abundant elements on Earth, significantly reducing raw material costs compared to lithium. This makes sodium-ion batteries potentially cheaper to produce.
- Safety: Sodium-ion batteries are less prone to thermal runaway and overheating, which enhances their safety profile, particularly in large-scale applications.
- Environmental Impact: Sodium-ion batteries are more environmentally friendly, as sodium extraction has a lower environmental impact than lithium mining. Additionally, they are easier to recycle.
- Performance at Low Temperatures: Sodium-ion batteries tend to perform better at lower temperatures compared to lithium-ion batteries, making them suitable for applications in colder climates.
Sodium-ion Battery Cons
- Lower Energy Density: Sodium-ion batteries typically have a lower energy density than lithium-ion batteries, meaning they store less energy per unit of weight. This can limit their use in applications where size and weight are critical, such as portable electronics and high-performance electric vehicles.
- Heavier and Bulkier: The larger ionic radius of sodium than lithium results in heavier and bulkier batteries, which can be a drawback for certain applications.
- Limited Commercial Availability: Despite significant advancements, sodium-ion batteries are still in the development phase and are not yet widely available on the commercial market. Scaling up production remains a challenge.
Part 8. Where sodium-ion batteries make the most sense
You might consider sodium-ion batteries if you are working with:
- Energy storage systems
- Solar storage
- Backup power systems
- Low-speed electric vehicles
- Telecom base stations
In these applications, cost, safety, and long cycle life often matter more than maximum energy density.
Part 9. Challenges of sodium-ion battery technology
Despite the advantages, sodium-ion batteries still face several technical challenges:
- Lower energy density
- Larger ionic radius affecting electrode stability
- Limited commercial supply chain (still developing)
However, these challenges are improving quickly as material science and manufacturing mature.
Part 10. Will sodium-ion battery replace lithium-ion battery?
The question of whether sodium-ion batteries will replace lithium-ion batteries is complex. Sodium-ion batteries offer significant advantages in terms of cost, safety, and environmental impact. However, their lower energy density means that they may not be suitable for all applications.
1. Reasons for Replacement
- Cost-Effectiveness: Sodium-ion batteries are cheaper to produce due to the abundance of sodium, making them an attractive option for cost-sensitive applications.
- Safety: Enhanced safety features make sodium-ion batteries suitable for applications where thermal stability is crucial.
- Sustainability: More environmentally friendly production and easier recycling processes align with the growing demand for sustainable technologies.
2. Limitations
- Energy Density: The lower energy density of sodium-ion batteries means that they are less suitable for high-energy applications, such as smartphones and high-performance electric vehicles.
- Commercialization: The technology is still in the development phase, and widespread commercial availability is limited. Overcoming production and scalability challenges will be crucial for broader adoption.
In summary, sodium-ion batteries are likely to complement rather than completely replace lithium-ion batteries. They may find their niche in applications where cost and safety are more critical than energy density.
Part 11. The future of sodium-ion batteries
Sodium-ion batteries are not here to replace lithium-ion batteries — they’re here to expand the battery ecosystem.
In the coming years, you’ll likely see:
- Hybrid lithium-sodium battery systems
- Sodium-ion grid storage expansion
- Low-cost EV adoption
- Improved high-energy sodium chemistries
If you’re planning long-term battery sourcing or product development, sodium-ion batteries are worth serious consideration.
Part 12. FAQs
1. Are sodium-ion batteries safer than lithium-ion batteries?
In many cases, yes. Sodium-ion batteries usually have better thermal stability and lower risk of overheating. This makes them attractive for energy storage, backup power, and industrial applications where safety is important.
2. Do sodium-ion batteries work well in cold weather?
Sodium-ion batteries generally perform better at low temperatures than lithium-ion batteries. This makes them suitable for outdoor systems, telecom equipment, and cold-climate energy storage.
3. How long do sodium-ion batteries typically last?
Most sodium-ion batteries offer around 2,000 to 5,000 cycles, depending on materials and operating conditions. In stationary storage, this often translates to many years of reliable use.
4. Are sodium-ion batteries cheaper than lithium-ion batteries?
They have the potential to be cheaper because sodium is abundant and widely available. As production scales up, the cost advantage is expected to become more noticeable.
5. Can sodium-ion batteries be used in electric vehicles?
Yes, but mainly in low-speed vehicles or cost-sensitive EVs. Their lower energy density makes them less suitable for long-range electric vehicles at the moment.
6. Are sodium-ion batteries environmentally friendly?
Sodium-ion batteries are often considered more sustainable because they rely on abundant materials and can reduce dependence on lithium, cobalt, and nickel.
Related Tags:
More Articles

Do Lithium Ion Batteries Have Nickel?
Do lithium ion batteries contain nickel? Learn which battery types use nickel, why it matters for energy density, and when LFP is a better choice.
Lithium Iron Phosphate vs Lithium Cobalt Oxide: Which is Better for Your Needs?
Compare LiFePO4 vs LiCoO2 batteries. Learn differences in safety, lifespan, cost, and energy density for EVs, storage, and electronics.
How Thin Can a Lithium Battery Get? Slim Battery Guide
Explore how slim lithium batteries work, how thin they can be, and what to consider before choosing a thin lithium battery.
How to Care for LiPo Batteries (2026 Guide): Charging, Storage & Lifespan Optimization
Learn how to care for LiPo batteries with proven charging, storage, and temperature practices. Improve LiPo battery lifespan, safety, and performance.
Best Rechargeable Batteries 2026: Tested by Type & Real Use Cases
Compare the best rechargeable batteries by type, lifespan, and performance. Tested lithium and NiMH picks for real-world use cases.