- Key takeaways
- Part 1. What is internal resistance in a lithium battery?
- Part 2. Internal resistance of an ideal cell
- Part 3. What causes battery internal resistance
- Part 4. How to measure internal resistance of a battery
- Part 5. AC method vs DC method: what professionals actually use
- Part 6. 18650 internal resistance: what normal values look like
- Part 7. Common mistakes when measuring internal resistance
- Part 8. Ways to reduce internal resistance in real-world use
- Part 9. FAQs
Key takeaways
- Battery internal resistance is one of the most important indicators of battery health, performance, and safety.
- You can measure it using simple DC load methods or more advanced AC impedance techniques, depending on accuracy needs.
- Even small increases in internal resistance can significantly affect voltage stability and heat generation.
- 18650 cells vary widely in internal resistance depending on chemistry, age, and usage conditions.
- If you understand how to measure it correctly, you can quickly judge whether a battery is still reliable or already degrading.
Part 1. What is internal resistance in a lithium battery?
In simple terms, internal resistance refers to the opposition to the flow of electrical current inside the battery. Just like any electrical circuit, a battery has resistance that slows down or limits the movement of charge. This resistance can affect various aspects of battery behavior, such as its efficiency and its ability to deliver power when needed.
Internal resistance in lithium batteries is made up of two primary components: ohmic resistance and polarization resistance.
-
Ohmic Resistance: This type of resistance is caused by the physical materials inside the battery. These materials include the electrodes, electrolyte, and separator. The resistance here is constant and does not vary with current or temperature, and it is mainly a result of the materials’ inherent properties.
-
Polarization Resistance: Polarization resistance, on the other hand, is more dynamic. It occurs due to the electrochemical reactions inside the battery, which create a temporary barrier (polarization) that impedes the flow of electrons. This resistance can vary depending on factors like the charge or discharge rate, the battery’s age, and temperature.
Understanding these two components of internal resistance helps explain how and why it affects battery performance.
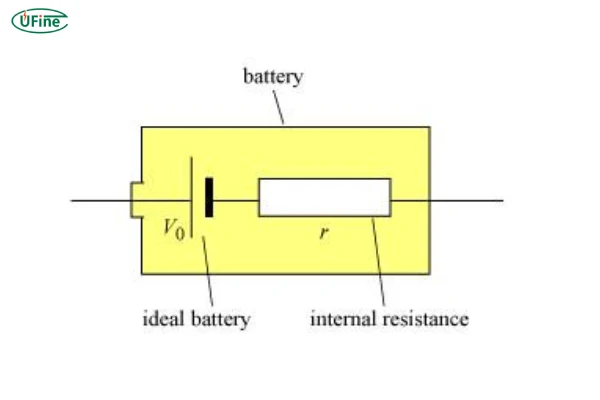
Part 2. Internal resistance of an ideal cell
In textbooks, you’ll often see a simplified idea: the internal resistance of an ideal cell is zero.
That sounds clean and perfect—but in reality, it’s more of a theoretical reference point than something that actually exists.
An ideal battery would:
- Deliver 100% of its stored energy to the load
- Produce no heat during discharge
- Maintain constant voltage regardless of current
But the moment you move from theory to real chemistry, things change.
Inside a real battery, electrons don’t flow freely without obstacles. They interact with electrode materials, electrolyte ions, and internal interfaces. All of this creates resistance—small, but unavoidable.
So instead of thinking “why isn’t it zero?”, it’s more accurate to think:
every real battery is a balance between energy storage and internal loss.
That’s why even brand-new lithium cells always show measurable internal resistance, no matter how good the design is.
Part 3. What causes battery internal resistance
Once you understand that internal resistance is unavoidable, the next question becomes more practical: where does it actually come from?
In most lithium-ion batteries (including 18650 cells), internal resistance is not a single thing—it’s a combination of several small resistive effects happening at different levels.
The main contributors are:
- Electrolyte resistance – ions moving through the liquid or gel electrolyte
- Electrode resistance – electron flow through active materials and current collectors
- Interface resistance – reactions happening at electrode-electrolyte boundaries
- Separator resistance – physical barrier adding slight ionic resistance
- Temperature effects – cold conditions slow ion movement and increase resistance
You don’t usually see these individually, but together they define how “easy” it is for the battery to deliver power.
To make it more intuitive, here’s a simple breakdown:
| Source of resistance | What it feels like in real use |
|---|---|
| Electrolyte limits | Slight delay in power delivery |
| Electrode structure | Reduced high-current performance |
| Interface reactions | Gradual aging over time |
| Low temperature | Weak output in cold weather |
What’s important here is that internal resistance is not static. It evolves as the battery ages, cycles, and experiences different temperatures.
That’s also why two identical batteries can behave very differently after a few months of use.
Part 4. How to measure internal resistance of a battery
If you’re wondering how to measure internal resistance of a battery in a practical way, the good news is: you don’t always need lab equipment.
The most common field method is the DC load method, and it’s surprisingly straightforward.
Here’s the logic behind it:
You measure the voltage before and after applying a known load, then calculate the drop.
A basic formula used in practice:
𝑅=Δ𝑉/𝐼
𝑉=𝐼𝑅=12V=IR=12
Where:
- ΔV = voltage drop under load
- I = discharge current
To make it clearer, here’s how the process usually goes:
- Measure the open-circuit voltage (no load)
- Apply a known load or discharge current
- Measure voltage again under load
- Calculate internal resistance using the formula
This method is widely used because it’s fast and doesn’t require expensive tools. However, it’s not perfect—especially for high-precision testing.
Part 5. AC method vs DC method: what professionals actually use
When people talk about how to measure battery resistance, they usually refer to two main approaches: DC and AC.
Quick Evaluation: DC Methods
If you need a fast and simple evaluation of internal resistance, DC methods like the short-circuit current or pulse current method are suitable. These are commonly used in mass production environments for preliminary testing.
In-depth Analysis: AC Methods
For more accurate results and a detailed analysis of battery health, AC methods, especially Electrochemical Impedance Spectroscopy (EIS), are the best choice. These methods are commonly used in research, development, and high-quality control applications where precision is critical.
| Method | How it works | Accuracy | Typical use case |
|---|---|---|---|
| DC method | Measures voltage drop under load | Medium | DIY testing, field checks |
| AC impedance (EIS) | Uses alternating signal response | High | Labs, manufacturing |
The AC method (often called impedance spectroscopy) is what professional labs use. It can capture very small changes in resistance that DC methods might miss.
It’s not necessary for everyday use, but it explains why high-end battery testing equipment is so expensive.
At Ufine Battery, as a leading lithium battery manufacturer in China, we take great care to minimize internal resistance in our products. Through advanced production processes and the use of high-quality materials, we ensure that our batteries maintain low internal resistance, which results in better performance and longer life for our clients.
VUfine Battery can customize capacity, voltage, discharge rate, and dimensions to match your exact application needs — from prototype development to large-scale production.
Get Your Custom Solution TodayPart 6. 18650 internal resistance: what normal values look like
If you’re working with lithium-ion cells, especially cylindrical ones, 18650 internal resistance is one of the most important health indicators.
Fresh high-quality 18650 cells usually have very low resistance. But over time, that value increases as the battery ages.
Here’s a realistic reference range:
| Condition | Internal resistance (typical) |
|---|---|
| New high-quality cell | 15–30 mΩ |
| Normal used cell | 30–60 mΩ |
| Aged or degraded cell | 60–100+ mΩ |
Capacity might still look “okay” even when resistance increases. But under load, you’ll feel the difference immediately—less power, more heat, and earlier cutoff.
That’s why professionals often say: capacity tells you how much energy is inside, but internal resistance tells you how usable that energy really is.
Part 7. Common mistakes when measuring internal resistance
Even though the process sounds simple, people often get misleading results because of small mistakes.
Here are a few things that quietly affect accuracy:
- Measuring immediately after charging (voltage instability)
- Using unstable or inconsistent load currents
- Ignoring temperature effects (cold batteries show higher resistance)
- Using low-quality multimeters that can’t capture fast voltage changes
These factors might not seem dramatic, but they can easily skew results enough to make a good battery look bad—or the opposite.
Part 8. Ways to reduce internal resistance in real-world use
Now, you might wonder: can you actually reduce internal resistance?
The honest answer is: not permanently, once it’s aged—but you can slow down the increase.
In real usage, the most effective habits are:
- Avoid deep discharges whenever possible
- Keep batteries away from high heat
- Don’t push high current loads continuously
- Store batteries at moderate charge levels
Over time, internal resistance naturally increases due to chemical changes inside the cell. You can’t stop it completely, but you can definitely slow it down.
Part 9. FAQs
1. What is the difference between internal resistance and external load?
Internal resistance is inside the battery, while external load is the device using the power. Internal resistance causes voltage drop before energy even reaches your device.
2. Is internal resistance the same for all battery types?
No. Lithium-ion, LiFePO4, NiMH, and lead-acid batteries all have different baseline internal resistance ranges and behavior.
3. What tools are best for accurate internal resistance measurement?
Professional battery analyzers and impedance testers are most accurate, while multimeters and simple load testers provide basic estimates.
4. Can internal resistance be measured during charging?
It is not recommended. Charging conditions distort voltage readings and lead to inaccurate resistance calculations.
5. Why do two identical batteries show different internal resistance?
Even identical models vary due to manufacturing differences, aging history, storage conditions, and usage patterns.
Related Tags:
More Articles

The Ultimate Guide to Small Size Battery
Learn small battery types, sizes, and applications. Compare lithium, alkaline, and coin cells. Find the best small size battery for your device or project.
Lithium Battery Comparison: Soft Pack Vs. Hard Pack
Compare soft pack vs hard pack lithium battery. Understand structure, safety, energy density, and applications to choose the right battery.
What Size are 18650 Batteries?
Learn exact 18650 battery dimensions (18mm × 65mm), tolerances, and pack configurations. Compare 18650 size with 21700 and others for better battery selection.
The Ultimate Guide to Battery Terminal Clamps
Learn battery terminal clamps, including types, materials, installation, and how to choose the right battery clamp for car and industrial use.
What Is a Lithium-Ion Battery Pack?
Learn what a lithium ion battery pack is, how it works, key components, configurations, and real-world applications for industrial and commercial use.