- Key Takeaways
- Part 1. What is lithium?
- Part 2. Is lithium a solid, liquid, or gas?
- Part 3. Physical states of lithium
- Part 4. Why lithium is ideal for batteries
- Part 5. Where is lithium found?
- Part 6. How lithium is processed
- Part 7. Is lithium dangerous?
- Part 8. Other uses of lithium
- Part 9. FAQ: Is lithium a solid, liquid, or gas?
Lithium powers modern devices—from smartphones to electric vehicles. But a common question is: is lithium a solid, liquid, or gas?
Key Takeaways
- Lithium is a solid at room temperature and normal pressure.
- It becomes liquid at 180.5°C and gas at 1,342°C.
- Most applications use solid lithium compounds, not pure lithium metal.
- Lithium offers high energy density and low weight, ideal for batteries.
- Its high reactivity requires controlled environments and proper design.
Part 1. What is lithium?
Lithium (Li) is a chemical element with atomic number 3. It belongs to the alkali metal group and is known for its high reactivity.
Key characteristics:
- Lightest metal in the periodic table
- Soft, silvery-white appearance
- Reacts quickly with air and water
- Widely used in energy storage systems
Lithium is essential in battery manufacturing. For example, understanding its source and supply chain helps in material selection and cost control, as explained in this guide on where lithium batteries come from.
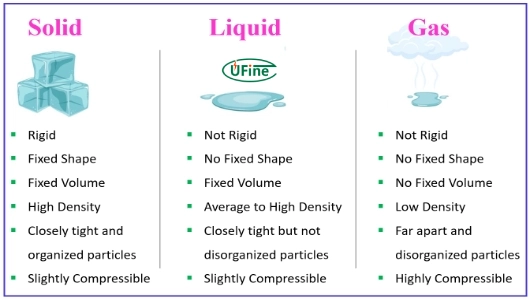
Part 2. Is lithium a solid, liquid, or gas?
Short Answer:
Lithium is a solid at room temperature.
Detailed Explanation
| State | Condition | Practical Meaning |
|---|---|---|
| Solid | ~20°C (room temp) | Normal state in nature and industry |
| Liquid | Above 180.5°C | Used in high-temperature systems |
| Gas | Above 1,342°C | Rare, mostly research use |
So:
- Is lithium a gas? → No, not under normal conditions
- Is lithium a liquid? → No, unless heated above its melting point
- Is lithium a solid liquid or gas? → It can exist in all three states, but is solid in everyday conditions
Part 3. Physical states of lithium
1 Solid lithium (primary state)
Lithium is almost always used in its solid form.
Key properties:
- Extremely low density (~0.534 g/cm³)
- High electrochemical potential
- Strong reactivity with water
When lithium reacts with water, it forms lithium hydroxide and hydrogen gas, a behavior documented by authoritative sources like Encyclopaedia Britannica’s lithium overview.
Engineering note:
In lithium-ion batteries, lithium exists in stable compounds such as:
- Lithium iron phosphate (LiFePO₄)
- Lithium cobalt oxide (LCO)
2 Liquid lithium (high-temperature state)
Lithium becomes liquid at 180.5°C.
Applications:
- Heat transfer fluids in advanced systems
- Alloy production for lightweight materials
Advantages:
- Efficient heat transfer
- High thermal conductivity
Limitations:
- Requires sealed and controlled environments
3 Gaseous lithium (extreme conditions)
Lithium turns into gas at 1,342°C.
This state is:
- Rare in industry
- Mainly used in laboratory and atomic research
Part 4. Why lithium is ideal for batteries
Lithium’s properties make it the top choice for modern batteries.
- High Energy Density
Lithium stores more energy per weight than most metals. - Lightweight
Ideal for: Electric vehicles, Portable electronics - High Voltage
Lithium-based cells provide higher output voltage. - Rechargeability
Lithium-ion batteries support long cycle life.
Battery voltage differences also affect performance and selection. For example, comparing common designs like 3.7V vs 3.8V lithium batteries helps optimize system design.
Part 5. Where is lithium found?
Lithium is not found in pure form due to its reactivity.
Main sources:
- Hard rock minerals (spodumene, petalite)
- Brine deposits (South America’s Lithium Triangle)
- Seawater (low concentration)
Global production data and reserves can be referenced from USGS lithium statistics.
Part 6. How lithium is processed
The production process includes:
- Extraction (mining or brine pumping)
- Refining into lithium carbonate or hydroxide
- Material conversion for battery use
Processing efficiency directly impacts battery cost and sustainability.
Part 7. Is lithium dangerous?
Lithium is safe in controlled systems but has risks.
Key risks:
- Reacts with water → hydrogen gas + heat
- Fire hazard if exposed to moisture
- Irritation from dust or compounds
Modern lithium batteries reduce these risks through:
- Battery Management Systems (BMS)
- Thermal control
- Protective circuit design
Part 8. Other uses of lithium
Lithium is also used in:
- Medicine (mood stabilization)
- Glass and ceramics (heat resistance)
- Lubricants (industrial grease)
- Aerospace (lightweight alloys)
Part 9. FAQ: Is lithium a solid, liquid, or gas?
Is lithium a solid, liquid, or gas at room temperature?
Lithium is a solid at room temperature and normal pressure.
Can lithium become a liquid?
Yes. Lithium melts at 180.5°C and becomes liquid under high heat.
Is lithium ever a gas?
Yes, but only at extremely high temperatures above 1,342°C.
Why do people ask “is lithium a gas”?
Because lithium is highly reactive, some assume it behaves like a gas. In reality, it is a solid metal under normal conditions.
Is lithium in batteries solid or liquid?
Lithium exists in solid electrode materials, while lithium ions move through a liquid or gel electrolyte inside the battery.
Related Tags:
More Articles
Carbon Battery Guide: Everything You Need to Know (2026)
What is a carbon battery? Learn carbon battery technology, advantages, vs lithium-ion comparison, applications, and real-world selection tips.
LiPo Battery Bulging? Here’s What You Must Know
Discover why lithium polymer batteries swell, the hidden risks, and simple ways you can prevent it from damaging your devices.
Sodium-Ion Battery Explained: Better Than Lithium?
What is a sodium-ion battery and how is it made? Learn benefits, costs, and real-world uses compared to lithium-ion technology.
LiFePO4 Prismatic Cells Comparison: Top Manufacturers & Best Use Cases (2026)
Compare LiFePO4 prismatic cells from top manufacturers. Explore specs, cycle life, and best use cases for EV and energy storage systems.
What is the KC Certification of Lithium Battery?
Understand KC certification for lithium batteries, including requirements, testing process, kc battery compliance, and how it compares with CE and UL.